what are the conductors?
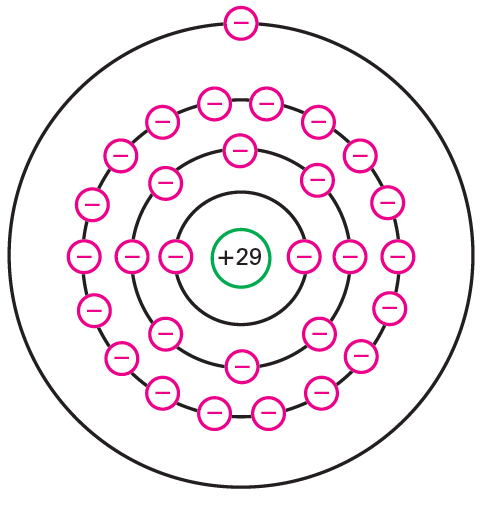
what are the properties of conductors in electrical terms? Copper is a decent conductor. The explanation is clear when we take a gander at its nuclear construction as shown in figure below. The core of the molecule contains 29 protons (positive charges). At the point when a copper particle has an impartial charge, 29 electrons (negative charges) circle the core like planets around the sun. The electrons travel in particular circles (moreover called shells). There are 2 electrons in the first circle, 8 electrons in the second, 18 in the third, and 1 in the external circle.
Stable Orbits
The positive core of Fig. above draws in the planetary electrons. The motivation behind why these electrons are not maneuvered into the core is the divergent (outward) power made by their round movement. This divergent power is actually equivalent to the internal pull of the core, so the circle is steady. The thought is like a satellite that circles the earth. At the correct speed and stature, a satellite can stay in a stable circle over the earth. The bigger the circle of an electron, the more modest the fascination of the core. In a bigger circle, an electron ventures all the more gradually, delivering less radiating power. The peripheral electron in above Fig. ventures gradually and feels basically no appreciation for the core.
What is the Core in electronics?
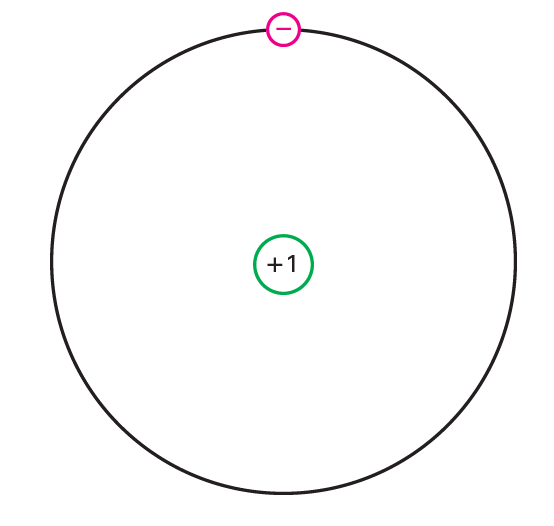
In hardware, the only thing that is important is the external circle. It is known as the valence circle. This circle controls the electrical properties of the atom. To underline the significance of the valence circle, we define the center of a particle as the core and all the internal circles. For a copper particle, the center is the core (129), and the first three circles (228). The center of a copper particle has a net charge of 11 since it contains 29 protons and 28 internal electrons. Figure below can help in imagining the center and the valence circle. The valence electron is in an enormous circle around a center that has a net charge of just 11. Along these lines, the internal draw felt by the valence electron is little.
Free Electron
Since the fascination between the center and the valence electron is powerless, an outside power can undoubtedly remove this electron from the copper molecule. This is the reason we frequently consider the valence electron a free electron. This is additionally why copper is a decent transmitter. The smallest voltage makes the free electrons flow from one particle to the following. The best transmitters are silver, copper, and gold.
Also read here:
https://eevibes.com/how-tunnel-diode-works/